Dr. Faizuddin Syed,
Professor in Mechanical Engineering,
DBS Institute of Technology,
Kavali, AP, India
Selecting a Suitable Electrode for Stainless Steels and Dissimilar Metal Joint
Stainless Steels are high Alloy Iron-base materials with large amount of Chromium. A small amount of Nickel and other alloying elements are also included. We need to consider some metallurgical facts when welding this type of metal group. Stainless steel have more enhanced resistance to fight with oxidation and corrosion. And also have improved physical properties at an elevated temp,. Chromium is the primary element, which is responsible for these properties. A maximum amount of 30% Chromium may be available in the Stainless steels.
The basic types of Stainless Steels are:
- Austenitic
- Martensitic
- Ferrite
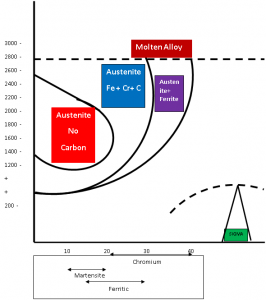
Martensite and Ferrite S Steels consists of different amounts of Chromium and Carbon. But Austenite can only exists in Carbon free Iron with up to 12% Cr.
But with the addition of Carbon, the Austenite (Gamma) loop extends up to 18%, depending on the amount of the Carbon added. The following diagram illustrates the Iron – Chromium Diagram and shows us the composition ranges for the Martensitic and Ferritic Steels.
There are some high Chromium low-carbon compounds called as Ferritic Stainless Steels, whose Ferrite phase is stable up to the melting point. These steels have no transformation face.
Austenitic Stainless Steels
A lot of fusion welding is carried out on this class of Stainless Steels. These steels are produced by adding the Alloy, which stabilize the Austenite at the range of all temperatures and eliminate the Austenite Ferrite Transformation. Austenitic S steel is basically an Iron-Chromium-Nickel Alloy group with different amounts of Carbon content and with some other elements, which provide special properties to resists corrosion and oxidation and can withstand at elevated temperatures. In fact these alloys never undergo the normal Austenite – Ferrite Transformation, by which they can’t get hardened by the heat treatment process. So this is the reason that they do not have hardened areas in the heat effected zones. These steels have excellent weldability.
Ferritic Stainless steels
These steels contains 17% to 27% of Chromium without significant amount of Carbon, Nickel, and other austenite formation elements. As per the following diagram the Ferrite phase exists up to the melting point and these steels can develop a little or no austenite on heating and no harden ability is developed. But some alloys like AISI 430 can develop a small amount of austenite at the grain boundaries and this will be converted to Martensites in the heat effected zone on cooling. The following table can provide you the composition of Ferritic Stainless steels. These alloys are subject to grain growth at a temperature above 2000F degrees. After cooling the heat effected zone adjacent to the fusion line contains large grains. But the size of the grains can’t be reduced by post heat treatment. The notch toughness can be increased during post weld annealing by the transformation of Martensite to Ferrite at the grain boundaries.
Composition of Ferritic Stainless Steels
| AISI | C% | Mn% | P% | S% | Si% | Cr% | Others |
| 405
430 430F 446 |
0.08 max
0.12 max 0.12 max 0.20 max |
1.00
1.00 1.25 1.50 |
0.040
0.040 0.06 0.040 |
0.030
0.030 0.15 min 0.030 |
1.00
1.00 1.00 1.00 |
11.50 -14.50
14.00-18.00 14.00-18.00 23.00-27.00 |
Al 0.10/0.30
————- Zr,Mo– 0.60 N-0.25 max |
Martensite Stainless Steels
These steels can be hardenable and this steel group is used for Cutlery etc., The following table shows the composition of these steels.
But more care must be taken in welding the Martensite steels, as these Martensite tends to produce in the heat effected zone only. So pre heating can slow down the cooling rate for Austenitie transformation and can control to minimum cracking.
Martensitic Stainless Steels Composition
| AISI | C max | MN max | P max | S max | Si max | Cr | Others |
| 403
410 414 416 420 431 440A 440B 440C |
0.15
0.15 0.15 0.15 0.15+ 0.20 0.60/0.75 0.75/0.95 0.95/1..20 |
1.00
1.00 1.00 1.25 1.00 1.00 1.00 1.00 1.00 |
0.040
0.040 0.040 0.06 0.040 0.040 0.040 0.040 0.040 |
0.030
0.030 0.030 0.15 min 0.030 0.030 0.030 0.030 0.030 |
0.50
1.00 1.00 1.00 1.00 1.00 1.00 1.00 1.00 |
11.5/13.00
11.5/13.50 11.5/13.50 12.0/14.00 12.0/14.00 15.0/17.00 16.0/18.00 16.0/18.00 16.0/18.00 |
—————
————— Ni:1.25-2.50 Zr,Mo-0.60+ ————— Ni.1.25/2.50 Mo.0.75 max Mo.0.75 max Mo.0.75Max |
These alloys are subject to grain growth a the temperatures of above 2000 F degrees. After cooing the heat effected zone adjacent to the fusion line can contain very large grains. The following figure is the heat effected zone of a welded joint of AISI 430 stainless steel with large grains and Intergranular Martensite. This grain size can’t be reduced significantly by post heat treatment The Notch toughness can be increased by post weld annealing by transforming Martensite to Ferrite at the grain boundaries. Ferritic Stainless steels filler metal can be used for welding these grades.
The Weld Metal and Delta Ferrite
The strength and the factors of corrosion and oxidation are necessary for the welding metal composition of stainless steels and it should match the base metal. Though Alpha ferrite is not found in these Austenite stainless steels, a small amount of Delta Ferrite can form , unless there is a sufficient quantity of Austenite stabilizing elements in the alloy. The alloys tending to stabilize Ferrite are Chromium, Molybdenum, Silicon and columbium. The past experience of stainless steel welding shows that, it is desirable that it should have a small amount of Delta Ferrite in the weld metal to prevent formation of hot cracks during cooling of the weld metal.
So welding electrode manufacturers keep and adjust 5% to 10% of Ferrite in the electrode chemical composition, so that the weld deposit should have this much of Ferrite.
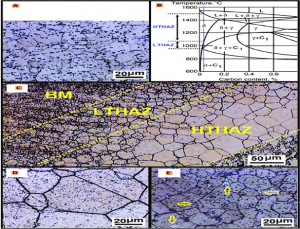
We can observes the structure of the HAZ , when carbon % and temperature is changed. The details are in the above figure:
A-is the Base metal microstructure of AISI 430 steel;
B-is the Fe-17% Cr-C phase diagram and HAZ of AISI 430 steel;
C-is the Microstructure gradient in the HAZ of Ferritic stainless steel;
D-is the grain growth and dispersion of carbide precipitates in HTHAZ;
E-is the Martensite formation, indicated by arrows, in LTHAZ.
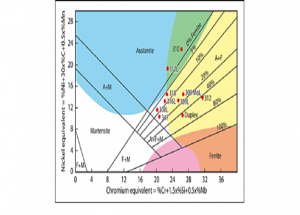
Schaeffler Diagram
We can predict the amount of Ferrite in the Schaeffler diagram, if we the composition of the composition of metal. This diagram is more useful to select and decide the type of the stainless steel welding electrode to be used for welding a difficult and dissimilar type of base materials.
Some difficult situations where the welded base material can un favorably effect the composition and can cause cracking problems. All these types of problems can be solved and quality welds may be created.
Schaeffler diagram with location of commonly used alloys
A-austenite; F-ferrite; M-martensite
Blue-hot cracking; yellow-sigma phase embritlement

Schaeffler diagram with location of commonly used alloys
A-austenite; F-ferrite; M-martensite
Blue-hot cracking; yellow-sigma phase embritlement
Which is a Correct and suitable electrode
for welding dissimilar base metals with SMAW process?
Selecting a correct and suitable electrode for welding an Alloy steel pipe with AISI 304 stainless steel pipe is as follows:
Chemical composition of Alloy steel pipe and AISI 304 stainless steel pipe
Base material – 1 : Alloy Steel pipe
Chemical composition: C – 0.13% ; Cr – 5.0%; Mo- 0.6%
Base material – 2 : AISI 304 Stainless Steel pipe
Chemical composition: C – 0.06% ; Cr – 18.0%; Ni – 8.0%; Mn – 2.0%; Si – 1%
Which is the best electrode to weld the above two materials ?
If we consider the dilution of 20% of base metal (10% from both the base metals) and 80% from welding electrode the result will be as follows:
Strength requirements, factors of corrosion and oxidation usually make it necessary for the weld metal composition of a stainless steel to closely match the base metal. It’s also desirable that a small amount of DELTA FERRITE should be present in the weld metal.
Trail No.1
Choose an electrode E308 to weld this joint.
Chemical composition of E308:
____________________________________________________________
C Cr Ni Mn Si P S
0.05% 19.0% 9.0% 2.5% 0.90% 0.04% 0.03%
____________________________________________________________
C Cr Mo Ni Mn Si
10% from base metal -1 0.013 0.5 0.06 — — —
10% from base metal – 2 0.006 1.8 — 0.8 0.2 0.1
80% from electrode-E308. 0.040 15.2 — 7.2 2.0 0.72
_____________________________________
0.059 17.5 0.06 8.0 2.2 0.82
_____________________________________
Calculate the chromium equivalent and Nickel equivalent by applying the above values.
We can calculate the Chromium equivalent with the following formula is as follows:
Cr eq = %Cr + %Mo + %Si x 1.5 + %Cb x 0.5
Calculate the Ni equivalent
We can calculate the Nickel equivalent with the following formula is as follows:
Ni eq = %Ni + %C x 30 + %Mn x 0.5
Plot the values of both Cr eq and Ni eq on the Schaeffler diagram. Then we can find that the filler metal have structure of Austenite, Martensite and Ferrite.
This is undesirable, as Martensite can cause cracking. So don’t select the electrode E308.
Trail No.2
Choose an electrode E309 to weld this joint.
Chemical composition of E309 (major elements):
____________________________________________________________
C Cr Ni Mn Si P S
0.15% 25.0% 9.0%
____________________________________________________________
C Cr Mo Ni Mn Si
10% from base metal -1 0.013 0.5 0.06 — — —
10% from base metal – 2 0.006 1.8 — 0.8 0.2 0.1
80% from electrode-E309. 0.120 20.0 — 9.6 2.5 0.90
_____________________________________
0.139 22.3 0.06 10.4 2.7 1.0
_____________________________________
Calculate the chromium equivalent and Nickel equivalent by applying the above values
We can calculate the Chromium equivalent with the following formula is as follows:
Cr eq = %Cr + %Mo + %Si x 1.5 + %Cb x 0.5
Calculate the Ni equivalent
We can calculate the Nickel equivalent with the following formula is as follows:
Ni eq = %Ni + %C x 30 + %Mn x 0.5
Plot the values of both Cr eq and Ni eq on the Schaeffler diagram. Then we can find that the filler metal have structure of Austenite only, and it will have approximately 5% of Ferrite which is desirable for this joint.
So we can decide that the electrode E309 is a suitable electrode for welding this dissimilar metal joint.
We can find out a suitable electrode for any type of difficult weld or Stainless steel and dissimilar metal joint with this procedure.






